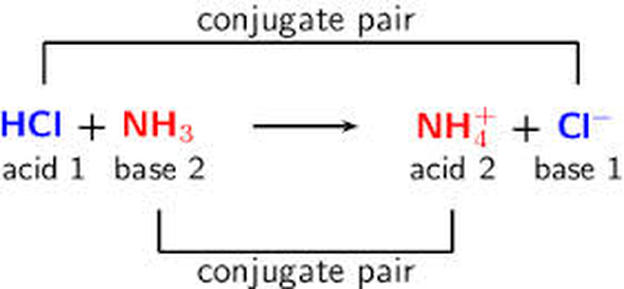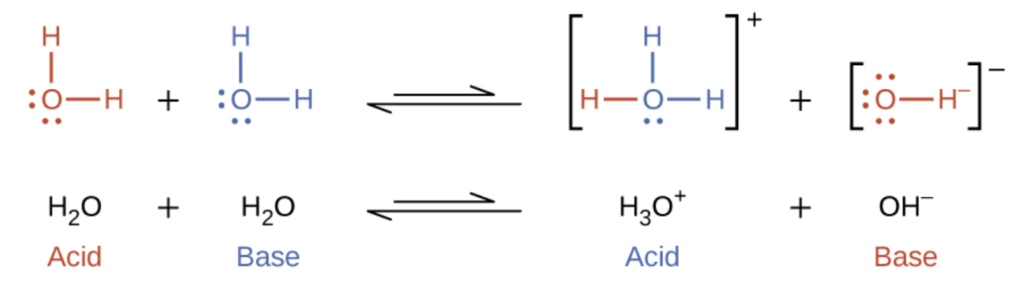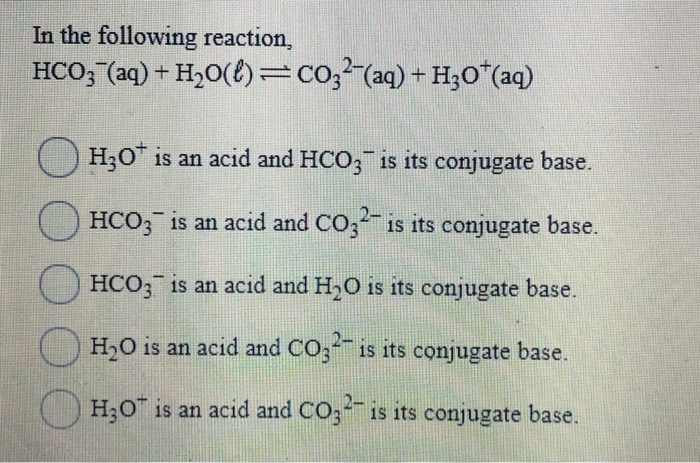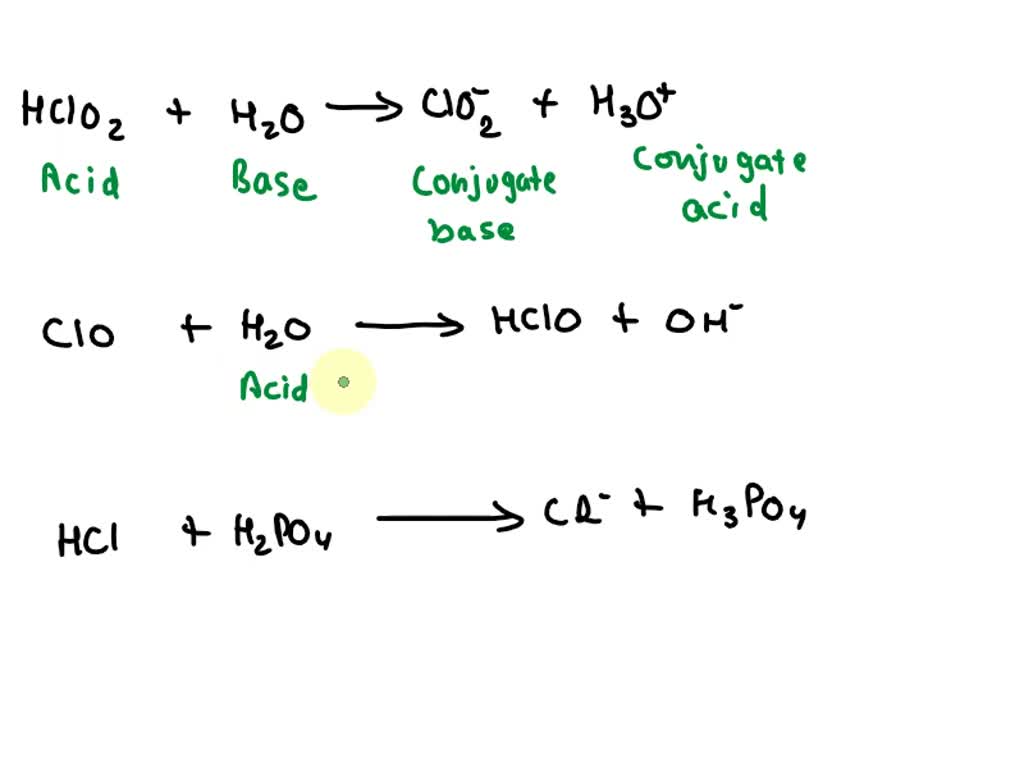
SOLVED: Complete the table Equation Acid Base Conjugate base Conjugate acid HClO2 + H2O → ClO−2 + H3O+ ClO− + H2O → HClO + OH− HCl + H2PO−4 → Cl− + H3PO4

SOLVED: QUESTION 11 In the following reaction: HCO3" (aq) + H2O (aq) -> H2CO3 (aq) + OH" (aq) 0 HCO3" is an acid and is H2CO3 its conjugate base. 0 H20 is

Réaction Acide-base. Réaction Chimique Neutralisant Les Propriétés Acides Et Basiques, Produisant Un Sel Et De L'eau. Utilisé Pour Déterminer Le Ph. Bronsted - Théorie De Lowry. Molécules De Hcl, Naoh, H2o Et

G) Sels Dans le chapitre 1, Acide + Base Sel + Eau HCl(aq) + NaOH(aq) NaCl(aq) + H2O Les sels sont des électrolytes forts qui se dissocient entièrement. - ppt video online télécharger